
Originally Posted by
Hoppalong

The group consists of yttrium and the 15 lanthanide elements (lanthanum, cerium, praseodymium, neodymium, promethium, samarium, europium, gadolinium, terbium, dysprosium, holmium, erbium, thulium, ytterbium, and lutetium).
Uses of Rare Earth Elements. Rare earth metals and alloys that contain them are used in many devices that people use every day such as computer memory, DVDs, rechargeable batteries, cell phones, catalytic converters, magnets, fluorescent lighting and much more.
The name rare earth is a historical misnomer, stemming from the fact that when they first discovered they were difficult to extract from surrounding matter. The USGS (United States Geological Survey) describes rare earth elements as moderately abundant, meaning that theyre not as common as elements like oxygen, silicon, aluminum, and iron (which together make up 90 percent of the Earths crust), but still well dispersed around the planet.
The rare earth element cerium, for example, is the 25th most abundant on Earth, making it about as common as copper. But unlike copper and similarly well-known elements, such as gold and silver, rare earths dont clump together in single-element lumps. Instead, because of their similar chemical composition (15 of the 17 rare earth elements occupy consecutive places on the periodic table), they bond freely with one another in minerals and clays.
As the academic David S. Abraham explains in his book on the topic, The Elements of Power, this makes for a grueling extraction process. To create rare earths from the ore that contains them the extracted material has to be dissolved in solutions of acids, over and over again, then filtered, and dissolved once more. The goal is not so much to remove rare earths from the mix as to remove everything else, writes Abraham.



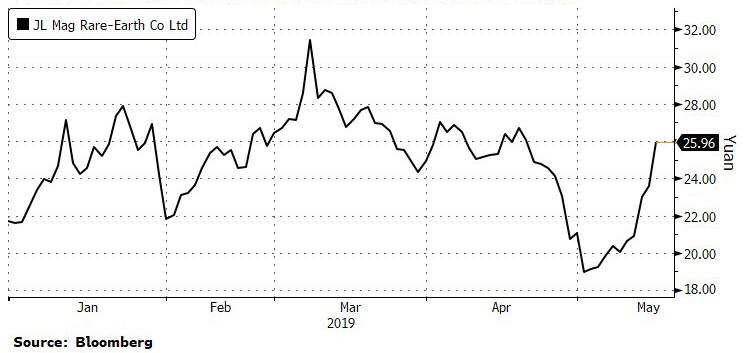
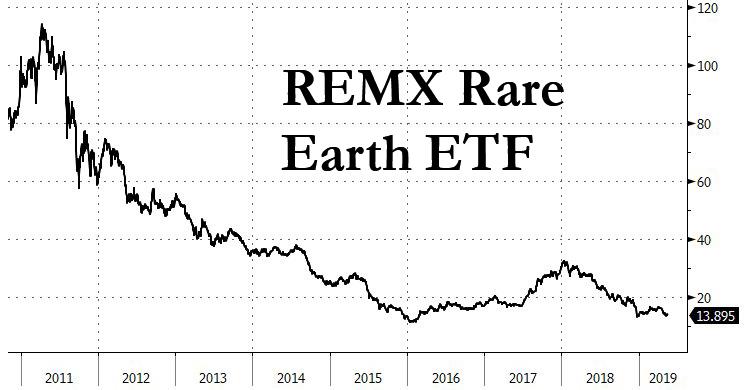

 Reply With Quote
Reply With Quote



